1. Hepatitis A
Sven Pischke, Heiner Wedemeyer
Epidemiology
Hepatitis A virus (HAV) infections occur worldwide, either sporadically or in epidemic outbreaks (Van Damme 2023). An estimated 1.4 million cases of HAV infections occur each year. HAV is usually transmitted and spread via the fecal-oral route (Lemon 1985). Thus, infection with HAV occurs predominantly in areas of lower socio-economic status and reduced hygienic standards, especially in developing, tropical countries. Not surprisingly, a study investigating French children confirmed that travel to countries endemic for HAV is indeed a risk factor for anti-HAV seropositivity (Faillon 2012). But hepatitis is not only a travel disease, but also endemic in countries of the western world. In industrialised countries like the US or Germany the number of reported cases has decreased markedly in the past decades, according to official data published by the Centers for Disease Control and Prevention (CDC, Atlanta, USA) and the Robert Koch Institute (RKI, Berlin, Germany) (Figure 1). This decrease is mainly based on improved sanitary conditions and anti-HAV vaccination. Vaccination programmes have also resulted in fewer HAV infections in various endemic countries including Argentina, Brazil, Italy, China, Russia, Ukraine, Spain, Belarus, Israel and Turkey (Hendrickx 2008).
Despite of the overall decrease in the frequency of hepatitis A in industrialised countries HAV outbreaks still occur. For example, HAV outbreaks have been described both in Europe and the US that were linked to frozen berries (Guzman Herrador 2014, Fitzgerald 2014) or imported pomegranate arils (Collier 2014).
HAV is transmitted fecal-orally either by person-to-person contact or ingestion of contaminated food or water. Usually HAV infection is restricted to humans and hepatitis A is no zoonosis. However, recently experimental HAV infection of pigs has been demonstrated (Song 2015, Migueres 2021).
Five days before clinical symptoms appear, the virus can be isolated from the feces of patients (Dienstag 1975). The hepatits A virus stays detectable in the feces up to two weeks after the onset of jaundice. Fecal excretion of HAV up to five months after infection can occur in children and immunocompromised persons. A recent study from Brasil evaluated the risk of household HAV transmission within a cohort of 97 persons from 30 families (Rodrigues-Lima 2013). Person-to-person transmission was seen in six cases indicating a relevant risk for relatives of patients with hepatitis A. On the other hand, there was no evidence of HAV transmission in another incident by an HAV-infected foodhandler in London (Hall 2014). Further studies are necessary to evaluate the use of HAV vaccination of relatives at risk in this setting.
Risk groups for acquiring an HAV infection in Western countries are health care providers, military personnel, psychiatric patients and men who have sex with men. Parenteral transmission by blood transfusion has been described but is a rare event. Mother-to-fetus transmission has not been reported (Tong 1981). Distinct genetic polymorphisms including variants in ABCB1, TGFB1, XRCC1 may be associated with a susceptibility to HAV (Zhang 2012).Regarding the severity of HAV infections an uncommon phenomenon has been described: it has been shown that the number of reported HAV infections in the USA decreased from 6 cases/ 100000 in 1999 to 0.4 cases/ 100000 in 2011 while the percentage of hospitalisations due to hepatitis A increased from 7.3% to 24.5% indicating that hepatitis A is getting rare but is causing still severe courses, particularly in elderly and patients with underlying liver diseases (Ly 2015).
Virology and diagnostics
Hepatitis A is an inflammatory liver disease caused by infection with the hepatitis A virus (HAV) (Van Damme 2023). HAV is a single-stranded 27 nm non-enveloped, icosahedral RNA virus, which was first identified by immune electron microscopy in 1973 (Feinstone 1973). The virus belongs to the hepadnavirus genus of the Picornaviridae. Recent structure-based phylogenetic analysis placed HAV between typical picornavirus and insect picorna-like viruses (Wang 2014). HAV uses host cell exosome membranes as an envelope which leads to protection from antibody mediated neutralisation (Feng 2013). Of note, only blood but not bile HAV shows host-derived membranes.
Seven different HAV genotypes have been described, of which four are able to infect humans (Lemon 1992).
The positive-sense single-stranded HAV RNA has a length of 7.5 kb and consists of a 5’ non-coding region of 740 nucleotides, a coding region of 2225 nucleotides and a 3’ non-coding region of approximately 60 nucleotides.
Acute hepatitis A is associated with a limited type I interferon response (Lanford 2011), which may be explained by cleavage of essential adaptor proteins by an HAV protease-polymerase precursor (Qu 2011). A dominant role of CD4+ T cells to terminate HAV infection has been established in HAV infected chimpanzees (Zhou 2012). However, in humans strong HAV-specific CD8 T cells have also been described, potentially contributing to resolution of infection (Schulte 2011). A failure to maintain these HAV-specific T cell responses could increase the risk for relapsing hepatitis A.
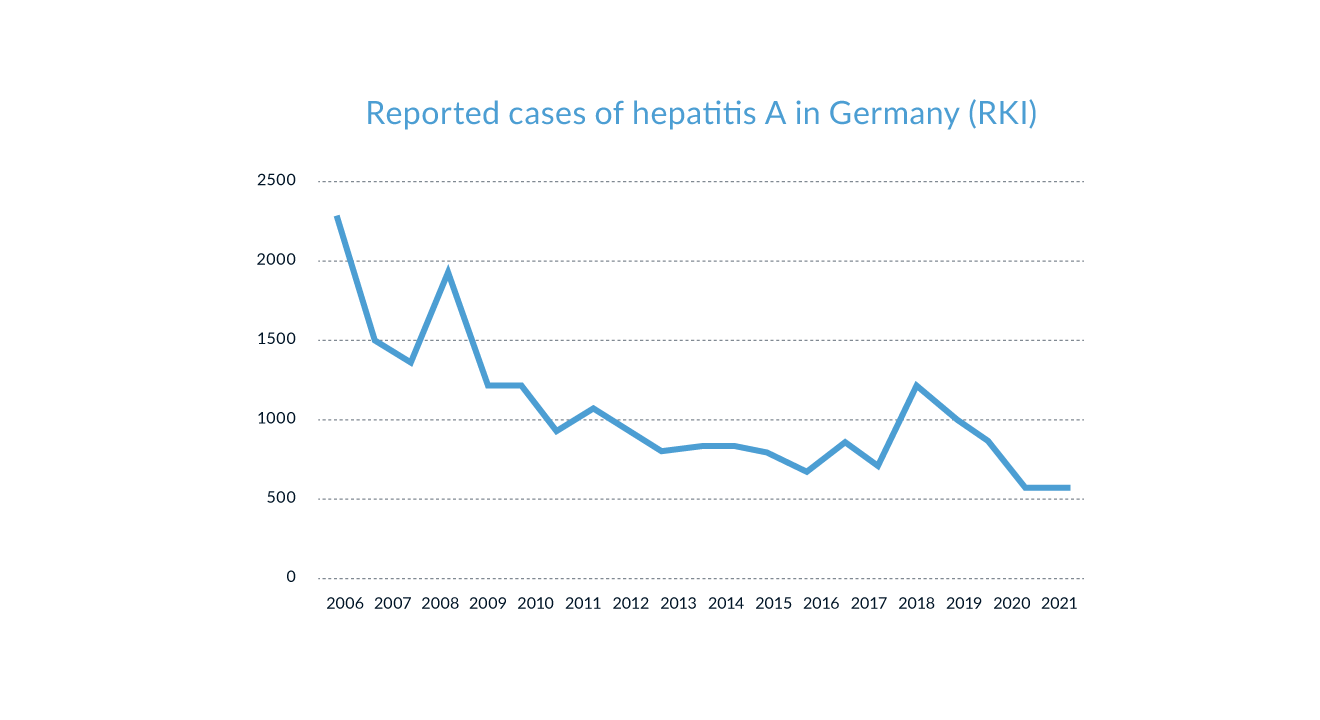 Figure 1. Number of reported cases of HAV infections in Germany over the last two decades (Source: Robert Koch Institut through 04/2023)
Figure 1. Number of reported cases of HAV infections in Germany over the last two decades (Source: Robert Koch Institut through 04/2023)
Diagnosis of acute HAV infection is based on the detection of anti-HAV IgM antibodies or HAV RNA. The presence of HAV IgG antibodies can indicate acute or previous HAV infection. HAV IgM and IgG antibodies also become positive early after vaccination, with IgG antibodies persisting for at least two to three decades after vaccination. Available serological tests show a very high sensitivity and specificity. Recently, a study from Taiwan revealed that HIV-infected patients develop protective antibody titres after HAV vaccination less frequently than healthy controls (Tseng 2012). In addition a study examining the immune response to HAV vaccination in 282 HIV-infected patients (Mena 2013) demonstrated that male sex or HCV coinfection were associated with lower response rates. The clinical relevance of these findings needs to be investigated in further studies.
A large study investigatied 183 adolescents (age 15- 16 years) who had initiated a two-dose HAV vaccination at age of 6, 12 or 15 months. Within these subgroups patients who got the vaccine during the earlier childhood at an age of 6 months and patients who got passively transferred maternal anti-HAV antibodies but were vaccinated at month 12 or 15 had a lower likelihood of carrying anti-HAV antibodies at the age of 15 or 16 years (Spradling 2015). This study demonstrates that HAV vaccination should not be vaccinated against HAV before 12 months of age, which is in line with the US recommendations. Delayed seroconversion may occur in immunocompromised individuals, and testing for HAV RNA should be considered in immunosuppressed individuals with unclear hepatitis. HAV RNA testing of blood and stool can determine if the patient is still infectious. However, it has to be kept in mind that various in-house HAV RNA assays may not be specific for all HAV genotypes and thus false negative results can occur.
Elevated results for serum aminotransferases and serum bilirubin can be found in symptomatic patients (Tong 1995). ALT levels are usually higher than serum aspartate aminotransferase (AST) in non-fulminant cases. Increased serum levels of alkaline phosphatase and gamma-glutamyl transferase indicate a cholestatic form of HAV infection. The increase and the peak of serum aminotransferases usually precede the increase of serum bilirubin. Laboratory markers of inflammation, like an elevated erythrocyte sedimentation rate and increased immunoglobulin levels, can also frequently be detected.
Recently within a small pilot study, examining 10 patients with acute hepatitis A, saliva of HAV infected patients has been shown to contain HAV RNA in 8/10 (80%) and anti HAV IgM in 10/10 (100%) (Armado Leon 2014). The relevance of this finding and the potential value of testing of saliva needs to be studied in larger cohorts.
Natural process and surveillance
The clinical course of HAV infection varies strongly, ranging from asymptomatic, subclinical infections to cholestatic hepatitis or life-threatening fulminant liver failure (Figure 2) (Van Damme 2023).
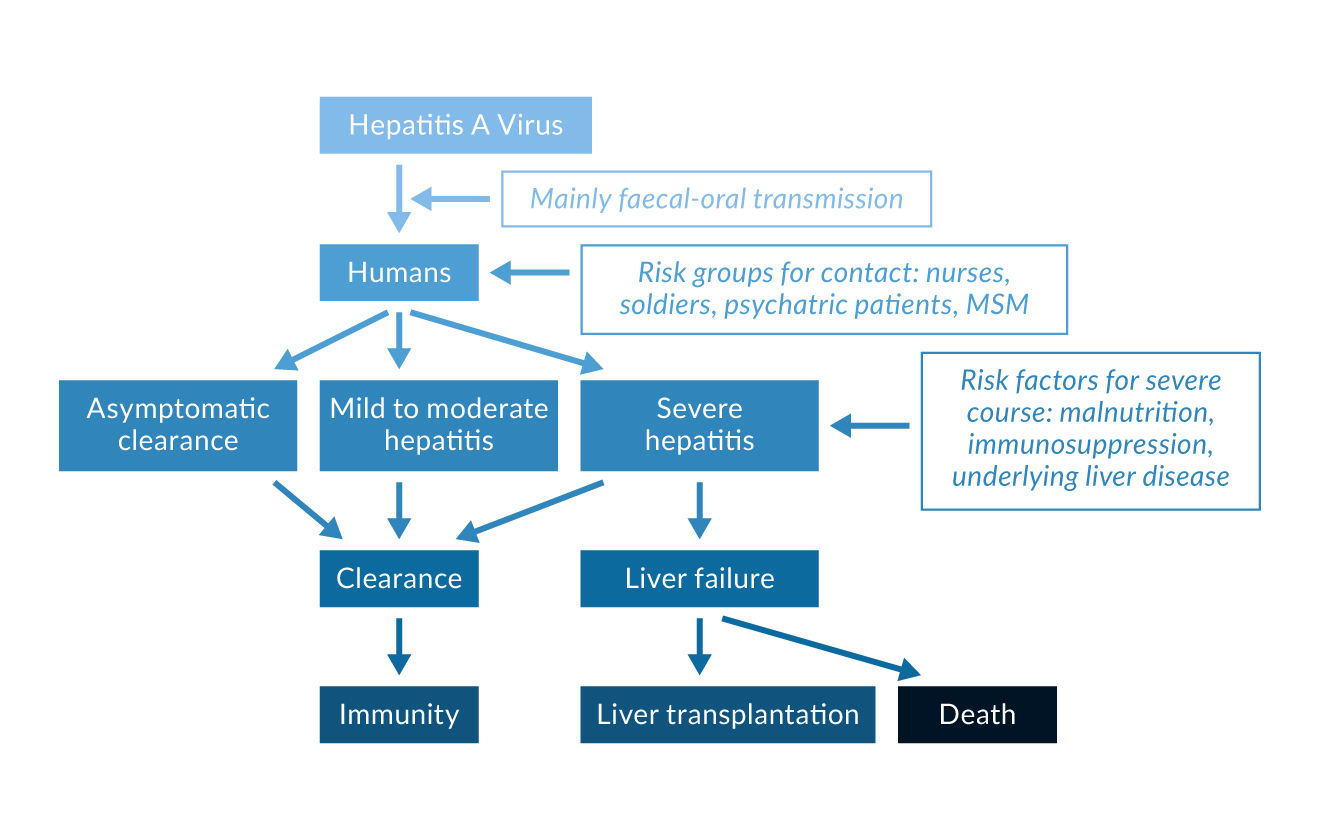 Figure 2. Possible courses of HAV infection
Figure 2. Possible courses of HAV infection
Most infections in children are either asymptomatic or unrecognised, while 70% of adults develop symptomatic hepatitis A with jaundice and hepatomegaly.
The incubation time ranges between 15 and 49 days with a mean of approximately 30 days (Koff 1992). Initial symptoms are usually non-specific and include weakness, nausea, vomiting, anorexia, fever, abdominal discomfort, and right upper quadrant pain (Lednar 1985). As the disease progresses, some patients develop jaundice, darkened urine, uncoloured stool and pruritus. The prodromal symptoms usually diminish when jaundice appears.
Approximately 10% of infections take a biphasic or relapsing course. In these cases the initial episode lasts about 3-5 weeks, followed by a period of biochemical remission with normal liver enzymes for 4-5 weeks. Relapse may mimic the initial episode of the acute hepatitis and complete normalisation of ALT and AST values may take several months. (Tong 1995). A recent investigation in two HAV-infected chimpanzees demonstrated that the CD4 count decreased after clinical signs of hepatitis A disappeared (Zhou 2012). Eventually an intrahepatic reservoir of HAV genomes that decays slowly in combination with this CD4 response may explain the second phase of disease, but further observations on human patients are required to verify this.
Cases of severe fulminant HAV infection leading to hepatic failure occur more often in patients with underlying liver disease (Patterson 2020). Conflicting data on the course of acute hepatitis A have been reported for patients with chronic hepatitis C. While some studies showed a higher incidence of fulminant hepatitis (Vento 1998), other studies do not confirm these findings and even suggest that HAV superinfection may lead to clearance of HCV infection (Deterding 2006). Other risk factors for more severe courses of acute hepatitis A are age, malnutrition and immunosuppression. Severity of liver disease during acute hepatitis A has recently been shown to be associated with a distinct polymorphism in TIM1, the gene encoding for the HAV receptor (Kim 2011). An insertion of 6 amino acids at position 157 of TIM1 leads to more efficient HAV binding and greater NKT lytic activity against HAV infected liver cells.
In contrast to hepatitis E, there are no precise data on the outcome of HAV infection during pregnancy. Some data suggest an increased risk of gestational complications and premature birth (Elinav 2006).
HAV infection has a lethal course in 0.1% of children, in 0.4% of persons aged 15-39 years, and in 1.1% in persons older than 40 years (Lemon 1985). In contrast to the other fecal-orally transmitted hepatitis, hepatitis E, no chronic courses of HAV infection have been reported so far.
In contrast to other hepatitis virus infections extrahepatic manifestations are really uncommon in HAV (Pischke 2007). If they occur, they usually show an acute onset and disappear upon resolution of HAV infection in most cases. Possible extrahepatic manifestations of acute HAV infection are arthralgia, diarrhoea, renal failure, red cell aplasia, generalised lymphadenopathy, and pancreatitis. Arthralgia can be found in 11% of patients with hepatitis A.
Very uncommon are severe extrahepatic manifestations like pericarditis and/or renal failure. An association of hepatitis A with cryoglobulinaemia has been reported but is a rare event (Schiff 1992). Furthermore, cutaneous vasculitis can occur. In some cases, skin biopsies reveal anti-HAV-specific IgM antibodies and complements in the vessel walls (Schiff 1992). In contrast to hepatitis B or C, renal involvement is rare, and there are very few case reports showing acute renal failure associated with HAV infection (Pischke 2007). Recently it has been shown that approximately 8% of hepatitis A cases are associated with acute kidney injury (Choi 2011).
Therapy
There is no specific antiviral therapy for treatment of hepatitis A (Migueres 2021). However a in vitro analysis of a cell culture system for HAV infection revealed efficient inhibition of HAV replication by cyclosporin A and silibinin (Esser-Nobis 2015). The clinical value of this in vitro observation still needs to be determined.
A study from the Netherlands investigated the use of post-exposure HAV vaccination or prophylaxis with immunglobulins in patients with household contact with HAV. In this study none of the patients who received immunoglobulins developed acute hepatitis A in contrast to some patients who received the vaccine. The study revealed that HAV vaccination post-exposure might be a sufficient option in younger patients (<40 years) while older patients (>40 years) might benefit from immunglobulins (Whelan 2013). The disease usually takes a mild to moderate course, which requires no hospitalisation, and only in fulminant cases is initiation of symptomatic therapy necessary. Prolonged or biphasic courses should be monitored closely. HAV may persist for some time in the liver even when HAV RNA becomes negative in blood and stool (Lanford 2011), which needs to be kept in mind for immunocompromised individuals. Acute hepatitis may rarely proceed to acute liver failure; liver transplantation is required in few cases. In the US, 4% of all liver transplantations performed for acute liver failure were due to hepatitis A (Ostapowicz 2002). In a cohort of acute liver failures at one transplant centre in Germany approximately 1% of patients suffered from HAV infection (Hadem 2008). The outcome of patients after liver transplantation for fulminant hepatitis A is excellent. Timely referral to liver transplant centres is therefore recommended for patients with severe or fulminant hepatitis A.
References
Armado Leon La, de Almeida AJ, de Paula VS et al. Longitudinal Study of Hepatitis A Infection by Saliva Sampling: The Kinetics of HAV Markers in Saliva Revealed the Application of Saliva Tests for Hepatitis A Study. PLOS One 2015; 10(12)
Centers for Disease Control and Prevention. Viral Hepatitis Surveillance – United States 2010. Hepatitis A virus, http://www.cdc.gov/hepatitis/Statistics/index.htm, accessed 26 January 2013.
Choi HK, Song YG, Han SH, et al. Clinical features and outcomes of acute kidney injury among patients with acute hepatitis A. J Clin Virol 2011;52:192-7.
Collier MG, Khudyakov YE, Selvage D, et al. Outbreak of hepatitis A in the USA associated with frozen pomegranate arils imported from Turkey: an epidemiological case study. Lancet Infect. Dis. 2014; 10: 976-981.
Deterding K, Tegtmeyer B, Cornberg M, et al. Hepatitis A virus infection suppresses hepatitis C virus replication and may lead to clearance of HCV. J Hepatol 2006;45:770-8.
Dienstag JL, Feinstone SM, Kapikian AZ, Purcell RH. Faecal shedding of hepatitis-A antigen. Lancet 1975;1:765-7.
Elinav E, Ben-Dov IZ, Shapira Y, et al. Acute hepatitis A infection in pregnancy is associated with high rates of gestational complications and preterm labor. Gastroenterology 2006;130:1129-34.
Esser-Nobis K, Harak C, Schult P, et al. Hepatology 2015, 62: 397-408
Faillon S, Martinot A, Hau I, et al. Impact of travel on the seroprevalence of hepatitis A in children. J Clin Virol. 2013 Jan;56(1):46-51. doi: 10.1016/j.jcv.2012.10.004.
Feinstone SM, Kapikian AZ, Purceli RH. Hepatitis A: detection by immune electron microscopy of a viruslike antigen associated with acute illness. Science 1973;182:1026-8.
Feng Z, Hensley L, McKnight KL, Hu F, Madden V, Ping L, et al. A pathogenic picornavirus acquires an envelope by hijacking cellular membranes. Nature. 2013;496(7445):367-71.
Fitzgerald M, Thornton L, O’Gorman J, et al. Outbreak of hepatitis A infection associated with the consumption of frozen berries, Ireland, 2013 - linked to an international outbreak. Euro Surveill. 2014;19(43):pii=20942.
Guzman-Herrador B, Jensvoll L, Einöder-Moreno M, et al. Ongoing hepatitis A outbreak in Europe 2013 to 2014: imported berry mix cake suspected to be the source of infection in Norway . Euro Surveill. 2014;19(15):pii=20775.
Hadem J, Stiefel P, Bahr MJ, et al. Prognostic implications of lactate, bilirubin, and aetiology in German patients with acute liver failure. Clin Gastroenterol Hepatol 2008;6:339-45.
Hall V, Abrahams A, Tubitt D, et al. No evidence of transmission from an acute case of hepatitis E in a foodhandler: follow-up of almost 1000 potentially exposed individuals, Lonndon, United Kingdom, April 2012Euro Surveill. 2014; 19 (30)
Hendrickx G, Van Herck K, Vorsters A, et al. Has the time come to control hepatitis A globally? Matching prevention to the changing epidemiology. J Viral Hepat 2008;15 Suppl 2:1-15.
Kim HY, Eyheramonho MB, Pichavant M, et al. A polymorphism in TIM1 is associated with susceptibility to severe hepatitis A virus infection in humans. J Clin Invest. 2011;121:1111-8.
Koff RS. Clinical manifestations and diagnosis of hepatitis A virus infection. Vaccine 1992;10 Suppl 1:S15-7.
Lanford RE, Feng Z, Chavez D, et al. Acute hepatitis A virus infection is associated with a limited type I interferon response and persistence of intrahepatic viral RNA. Proc Natl Acad Sci U S A 2011;108:11223-8.
Lednar WM, Lemon SM, Kirkpatrick JW, Redfield RR, Fields ML, Kelley PW. Frequency of illness associated with epidemic hepatitis A virus infections in adults. Am J Epidemiol 1985;122:226-33.
Lemon SM, Jansen RW, Brown EA. Genetic, antigenic and biological differences between strains of hepatitis A virus. Vaccine 1992;10 Suppl 1:S40-4.
Lemon SM. Type A viral hepatitis. New developments in an old disease. N Engl J Med 1985;313:1059-67.
Ly KN, Klevens RM. Trends in disease and complications of hepatitis A virus infection in the United States 199-2011: a new concern for adults. J. Infect. Dis. 2015, 58; 40-49
Mena G, García-Basteiro AL, Llupià A, Factors associated with the immune response to hepatitis A vaccination in HIV-infected patients in the era of highly active antiretroviral therapy. Vaccine. 2013;12;31(36):3668-74.
Migueres M, Lhomme S, Izopet J. Hepatitis A: Epidemiology, High-Risk Groups, Prevention and Research on Antiviral Treatment. Viruses. 2021;13(10):1900.
Ostapowicz G, Fontana RJ, Schiodt FV, et al. Results of a prospective study of acute liver failure at 17 tertiary care centers in the United States. Ann Intern Med 2002;137:947-54.
Patterson J, Hussey HS, Silal S, et al. Systematic review of the global epidemiology of viral-induced acute liver failure. BMJ Open. 2020;10(7):e037473.
Pischke S, Vogel A, Jaeckel E, Manns MP. Immunopathogenesis of Extrahepatic Manifestations in HAV, HBV, and HCV Infections. In: Liver Immunology. Totowa, New Jersey: Humana Press, 2007:209-17.
Rodrigues-Lima L, De Ameilda AJ, dos Santos Tourinho R, et al. Evidence of Hepatitis A Virus Person-to-Person Transmission in Household Outbreaks. PLOS One 2013, Vol. 9, Issue 7.
Qu L, Feng Z, Yamane, et al. Disruption of TLR3 signaling due to cleavage of TRIF by the hepatitis A virus protease-polymerase processing intermediate, 3CD. PLoS Pathog. 2011 Sep;7(9):e1002169.
Schiff ER. Atypical clinical manifestations of hepatitis A. Vaccine 1992;10 Suppl 1:S18-20.
Schulte I, Hitziger T, Giugliano S, et al. Characterisation of CD8+ T-cell response in acute and resolved hepatitis A virus infection. J Hepatol. 2011;54:201-8.
Song YJ, Park WJ, Park BJ, et al. J. Med. Virol. 2015, epub ahead of print
Spradling PR, Bulkow LR, Negus SE, et al. Persistence of seropositivity among persons vaccinated for hepatitis A during infancy by maternal antibody status: 15 year follow-up- Hepatology 2015, epub ahead of print
Tseng YT, Chang SY, Liu WC, et al. Comparative effectiveness of two doses versus three doses of hepatitis a vaccine in HIV-infected and HIV-uninfected men who have sex with men. Hepatology. 2012 Dec 19. doi: 10.1002/hep.26210. [Epub ahead of print]
Tong MJ, el-Farra NS, Grew MI. Clinical manifestations of hepatitis A: recent experience in a community teaching hospital. J Infect Dis 1995;171 Suppl 1:S15-8.
Tong MJ, Thursby M, Rakela J, McPeak C, Edwards VM, Mosley JW. Studies on the maternal-infant transmission of the viruses which cause acute hepatitis. Gastroenterology 1981;80:999-1004.
Van Damme P, Pintó RM, Feng Z, Cui F, Gentile A, Shouval D. Hepatitis A virus infection. Nat Rev Dis Primers. 2023;9(1):51.
Vento S, Garofano T, Renzini C, et al. Fulminant hepatitis associated with hepatitis A virus superinfection in patients with chronic hepatitis C. N Engl J Med 1998;338:286-90.
Wang X, Ren J, Gao Q, et al. Hepatitis A virus and the origin of picornaviruses. Nature 2014; epub ahead of print
Whelan J, Sonder GJ, Bovee L, et al. Evaluation of hepatitis A vaccine in post-exposure prophylaxis, The Netherlands, 2004-2012. PLoS One 2013, 17: e78914.
Zhou Y, Callendret B, Xu D, et al. Dominance of the CD4(+) T helper cell response during acute resolving hepatitis A virus infection. J Exp Med. 2012 Jul 30;209(8):1481-92. doi: 10.1084/jem.20111906. Epub 2012 Jul 2.
Zhang L, Yesupriya A, Hu DJ, et al. Variants in ABCB1, TGFB1, and XRCC1 genes and susceptibility to viral hepatitis A infection in Mexican Americans. Hepatology. 2012; 55:1008-18.
